
Glucose is one of your body's favorite sources of energy. Alpha and beta glucose are two common forms of the fuel that differ in their molecular structure and the types of nutrients they create.
What Is Glucose?
Video of the Day
Glucose molecules are simple sugars found in carbohydrate foods like fruits, vegetables, grains and dairy, according to the U.S. National Library of Medicine.
Video of the Day
After you eat, glucose travels through your blood to the rest of your body, where it breaks down to produce the energy you need to function, per a September 2020 StatPearls review. If there's any left over, your body stores it as glycogen until it's needed again.
Chemists use prefixes like alpha and beta to denote different versions of glucose — specifically, d-glucose, the most common form of the molecule, according to the American Chemical Society. To the uninitiated, these prefixes may seem mysterious, but the main differences have to do with their structure and the nutrients they comprise.
Alpha Glucose
Alpha glucose is the foundational molecule of starch, according to the University of California, Davis (UC Davis). It's a building block for foods like whole grains, beans, corn and potatoes, per the U.S. National Library of Medicine.
Beta Glucose
Beta glucose, on the other hand, is the molecular basis for a substance called cellulose, according to January 2014 research in The Arabidopsis Book. Cellulose is the main ingredient in plant cell walls, so you'll find it in fibrous foods like legumes, nuts and peas, per the 2020-2025 Dietary Guidelines for Americans.
Below, we'll go into more detail on the differences between alpha and beta sugars.
1. Chemical Structure
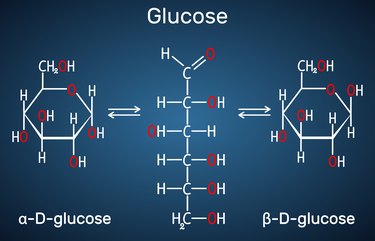
Every glucose structure — including alpha and beta glucose — contains six carbon atoms, 12 hydrogen atoms and six oxygen atoms (C6H12O6), according to StatPearls.
The structure of glucose includes a carbon backbone with hydrogen atoms (-H) and bonded hydrogen and oxygen atoms (-OH, or hydroxyl) attached to it. This network of atoms can either form a ring or a linear chain, though the ring form is more common, according to the University of Texas at Austin.
The difference between alpha and beta glucose is nothing more than the position of one of the -OH groups, according to UC Davis. If the -OH group to the right of the oxygen atom drawn in the upper-right corner of the hexagon is attached below the ring, the molecule is alpha glucose (shown above, at left). If the -OH group is above the ring, the molecule is beta glucose (shown above, at right).
2. Starch vs. Cellulose
Another distinction between alpha vs. beta glucose? The nutrients they make up.
Alpha glucose molecules link together to form starches. The 2020-2025 Dietary Guidelines for Americans recommends 5 cups of starchy vegetables like cassava, jicama and lima beans per week as part of a healthy diet.
Why? Your body breaks down starch more slowly than quick sources of energy like refined sugars, which means starchy foods can provide longer-lasting fuel, according to U.S. National Library of Medicine.
Beta glucose molecules link to form cellulose, which is the basis for dietary fiber, according to the March 2011 book Pulse Foods. The 2020-2025 Dietary Guidelines for Americans suggests between 22 and 28 grams of fiber daily for adults assigned female at birth, and between 28 and 34 grams of fiber daily for adults assigned male at birth. You can consume cellulose in foods like lentils, popcorn and raw fruits and vegetables.
Fiber is the part of plant food that your body can't digest or absorb, according to the Mayo Clinic. It thus passes through your intestines mostly intact, which supports digestive health and encourages normal bowel movements. It can also help keep your cholesterol and blood sugar levels in check, which may help prevent conditions like diabetes or high blood pressure, per the Mayo Clinic.
Related Reading
- StatPearls: "Physiology, Glucose"
- U.S. National Library of Medicine: "Carbohydrates"
- The Arabidopsis Book: "Cellulose Synthesis and Its Regulation"
- U.S. Department of Agriculture and U.S. Department of Health and Human Services: "Dietary Guidelines for Americans, 2020-2025"
- University of California, Davis: "Starch"
- University of Texas at Austin: "Molecule of the Day: Glucose"
- Pulse Foods: "Dietary fiber"
- Mayo Clinic: "Dietary fiber: Essential for a healthy diet"
- American Chemical Society: "D-Glucose"
- Northern Kentucky University: Tutorial -- Haworth Projections
Is this an emergency? If you are experiencing serious medical symptoms, please see the National Library of Medicine’s list of signs you need emergency medical attention or call 911.