Signs and Symptoms of Leaking Breast Implants
According to a June 2011 report by the U.S. Food and Drug Administration, an estimated 5 million to 10 million women worldwide have breast implants 2. Used for cosmetic reasons or to augment breast tissue after cancer surgery, these devices are filled with saline or silicone gel. Breast implants can rupture due to age, trauma, medical procedures or for other reasons 2. Signs and symptoms of breast implant leakage vary, depending on the type of device. Leaking implants typically require removal and replacement, if desired.
Loss of Volume
Saline-filled implants contain sterile salt water 6. If this type of implant ruptures, the fluid leaks out of the shell and is absorbed into the body. You will not experience symptoms from the leakage, but the implant will deflate and your breast will lose volume. The area around the deflated implant typically hardens and constricts over time, a condition called contracture, unless your surgeon removes the affected implant within the first 2 to 3 weeks. Loss of volume can also occur if a silicone breast implant ruptures and develops a small leak outside the shell.
- Saline-filled implants contain sterile salt water 6.
- The area around the deflated implant typically hardens and constricts over time, a condition called contracture, unless your surgeon removes the affected implant within the first 2 to 3 weeks.
Deformity
What Is the Difference Between a Regular Mammogram & a Bilateral Mammogram?
Learn More
Because the contents of a silicone gel-filled implant are thicker than the fluid in a saline-filled implant, it may not leak outside of a torn shell 6. Called a silent rupture, the leaking is not apparent to the woman or her doctor. On the other hand, if the silicone leaks outside the shell, you may notice a deformity in the size or shape of the breast due to the formation of scar tissue, or contracture.
- Because the contents of a silicone gel-filled implant are thicker than the fluid in a saline-filled implant, it may not leak outside of a torn shell 6.
- On the other hand, if the silicone leaks outside the shell, you may notice a deformity in the size or shape of the breast due to the formation of scar tissue, or contracture.
Local Signs and Symptoms
When a silicone implant ruptures and leaks outside the shell, you may notice changes in the tissue around the implant. Your skin may look red and inflamed, and the breast may look swollen. You may feel small fluid-filled lumps near the implant or notice discomfort in your shoulder or arm on the side of the leak. Your breast may feel numb or you may develop tingling, burning or pain in the affected area.
- When a silicone implant ruptures and leaks outside the shell, you may notice changes in the tissue around the implant.
- You may feel small fluid-filled lumps near the implant or notice discomfort in your shoulder or arm on the side of the leak.
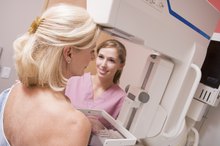
Diagnostic Imaging
What Are the Causes of Thickness in Breast Tissue?
Learn More
If your doctor suspects a silent rupture of a silicone gel-filled implant, imaging can help with the diagnosis 6. Magnetic resonance imaging of the breast can identify implant ruptures in up to 90 percent of cases, according to a July 2008 report published in "European Radiology." In contrast, mammograms can only identify ruptured implants approximately 25 percent of the time 25.
Silicone and Medical Conditions
The use of silicone gel-filled implants has raised concerns about the possibility of leaks causing serious medical conditions such as scleroderma, fibromyalgia and other connective tissue diseases 6. In fact, the FDA removed these devices from the market in February 1992 due to safety concerns. Two new types of silicone gel-filled implants were approved in 2006 6. After an extensive review of research studies related to potential complications of silicone gel-filled implants, the FDA concluded in 2011 that these devices are not associated with the development of breast cancer, connective tissue disease or problems with fertility and pregnancy 26.
Related Articles
References
- Institute of Medicine: Information for Women About the Safety of Silicone Breast Implants
- FDA: Breast Implants: Risks of Breast Implants
- Archives of Plastic Surgery: Augmentation Mammaplasty Using Implants -- A Review
- Oman Medical Journal: Breast Prothesis Leakage and Malignant Changes of the Breast
- European Radiology: Breast MRI -- Guidelines From the European Society of Breast Imaging
- FDA: FDA Update on the Safety of Silicone Gel-Filled Implants
- FDA.gov. What to know about breast implants. Updated October 23, 2019
- U.S. Food and Drug Administration. FDA update on the safety of silicone gel-filled breast implants. June 2011
- Balk EM, Earley A, Avendano EA, Raman G. Long-Term Health Outcomes in Women With Silicone Gel Breast Implants: A Systematic Review. Ann Intern Med. 2016;164(3):164-75. doi:10.7326/M15-1169
- Rocco N, Rispoli C, Moja L, et al. Different types of implants for reconstructive breast surgery. Cochrane Database Syst Rev. 2016;(5):CD010895. doi:10.1002/14651858.CD010895.pub2.
- Balk EM, Earley A, Avendano EA, Raman G. Long-Term Health Outcomes in Women With Silicone Gel Breast Implants. Annals of Internal Medicine. 2015;164(3):164. doi:10.7326/m15-1169
- FDA Update on the Safety of Silicone Gel-Filled Breast Implants. June 2011. Center for Devices and Radiological Health U.S. Food and Drug Administration.
- Rocco N, Rispoli C, Moja L, et al. Different types of implants for reconstructive breast surgery. Cochrane Database of Systematic Reviews. 2016. doi:10.1002/14651858.cd010895.pub2
Writer Bio
Marcy Brinkley has been writing professionally since 2007. Her work has appeared in "Chicken Soup for the Soul," "Texas Health Law Reporter" and the "State Bar of Texas Health Law Section Report." Her degrees include a Bachelor of Science in Nursing; a Master of Business Administration; and a Doctor of Jurisprudence.