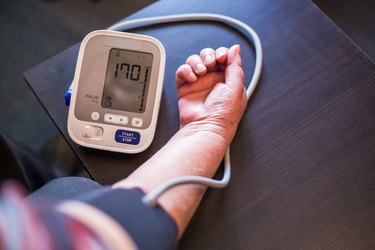
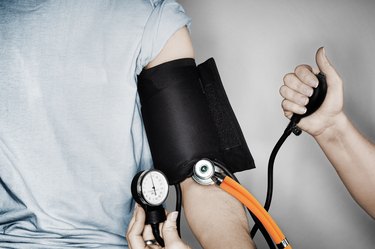
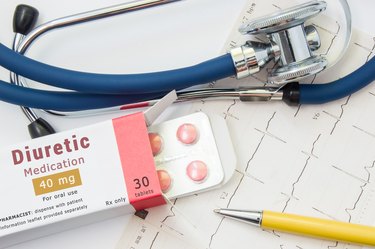
We understand the importance of maintaining a healthy heart and cardiovascular system. On our heart and cardiovascular health conditions page, we offer a wealth of resources to help you better understand various cardiovascular conditions, including high blood pressure, heart disease and stroke.
Our team of medical experts provides in-depth articles on symptoms, causes, diagnosis and treatment options for a wide range of conditions, as well as tips for maintaining heart health through lifestyle changes like exercise and healthy eating. We also offer advice on managing risk factors that can contribute to cardiovascular conditions, such as smoking, high cholesterol and diabetes.
Whether you are looking to improve your overall heart health or are dealing with a specific condition, our heart and cardiovascular health conditions page has something for you."